About
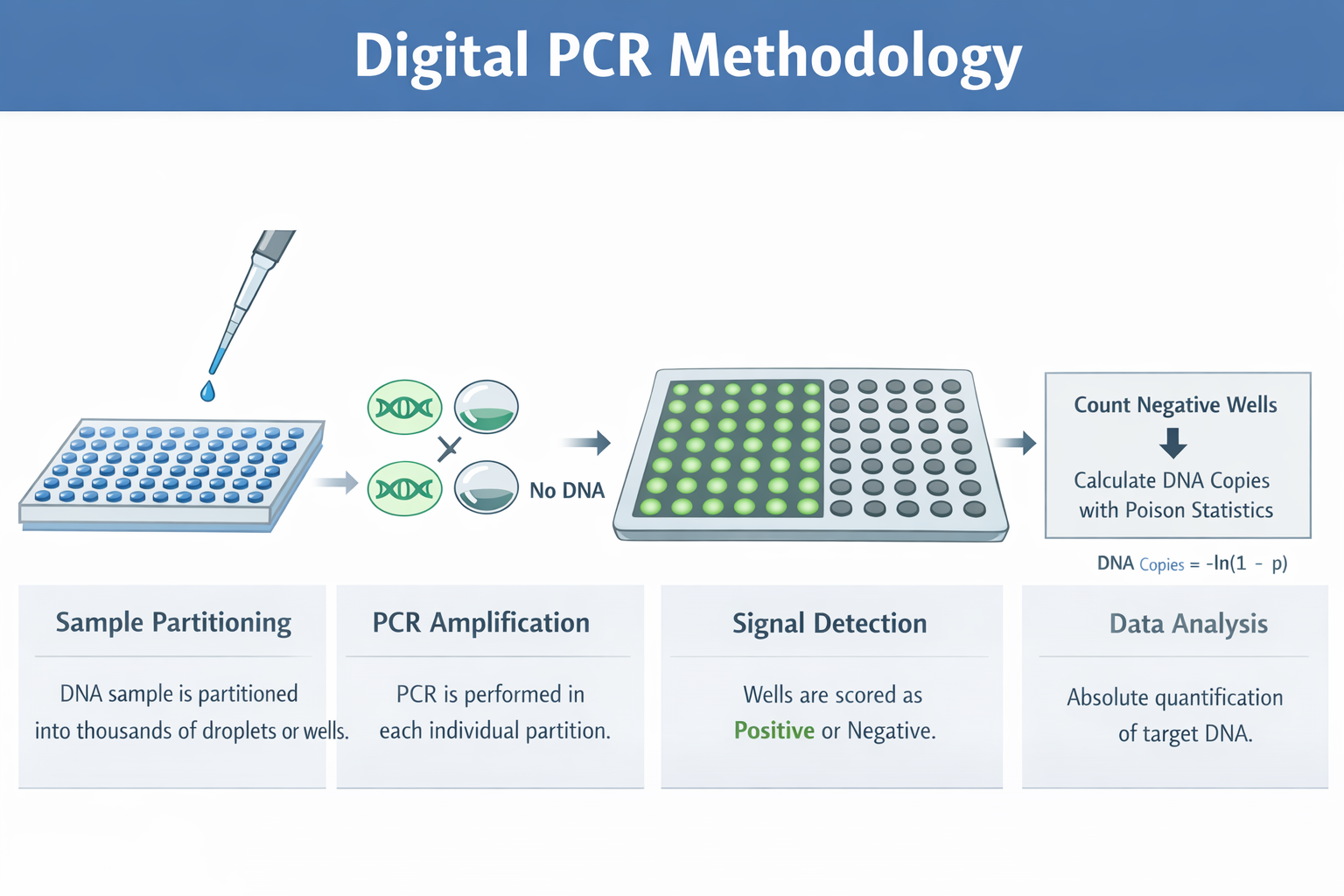
Digital PCR
Digital PCR enables precise, accurate quantification of plasmid DNA in drug products, improving assay accuracy and ensuring consistent vaccine development pipelines.

Project details
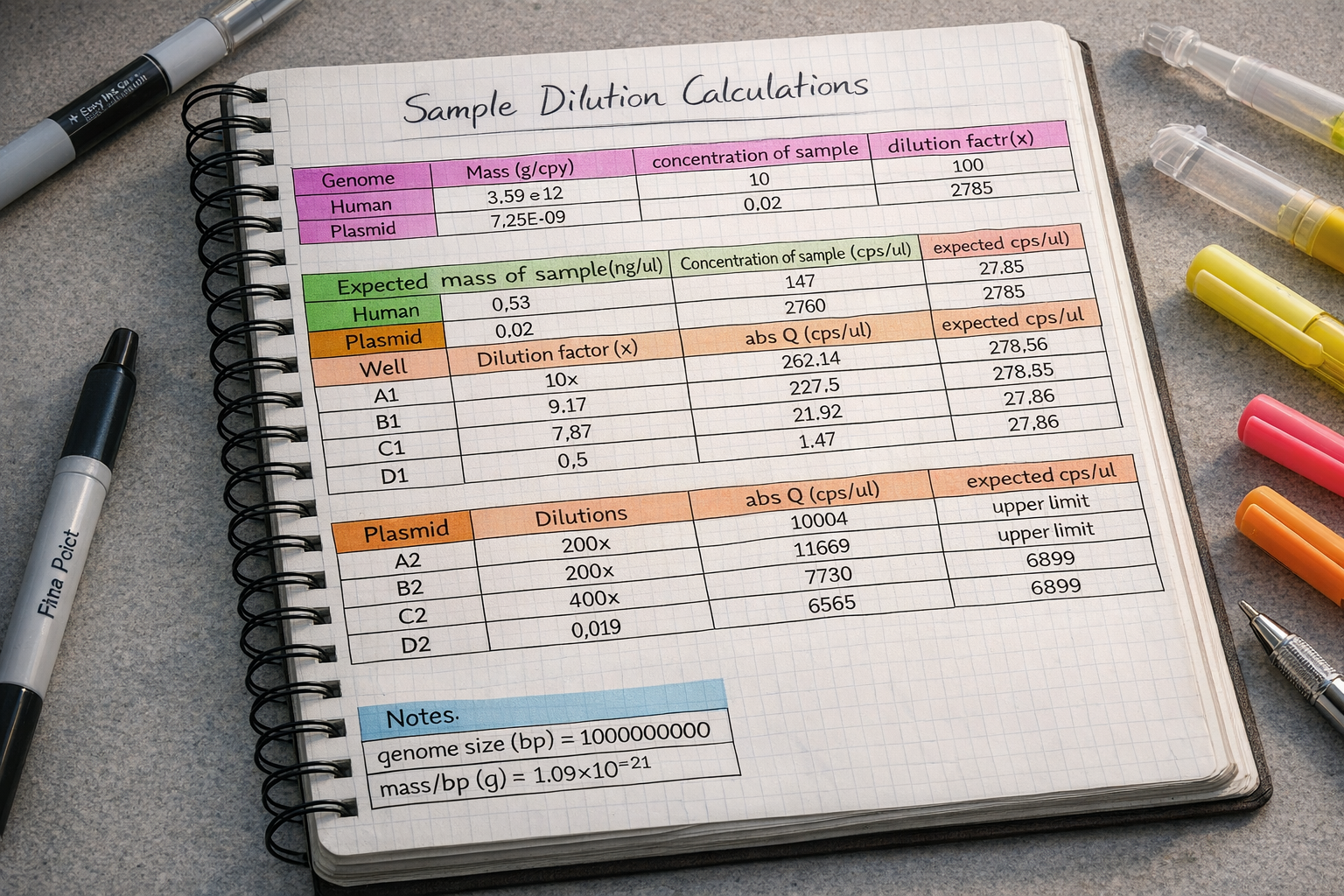
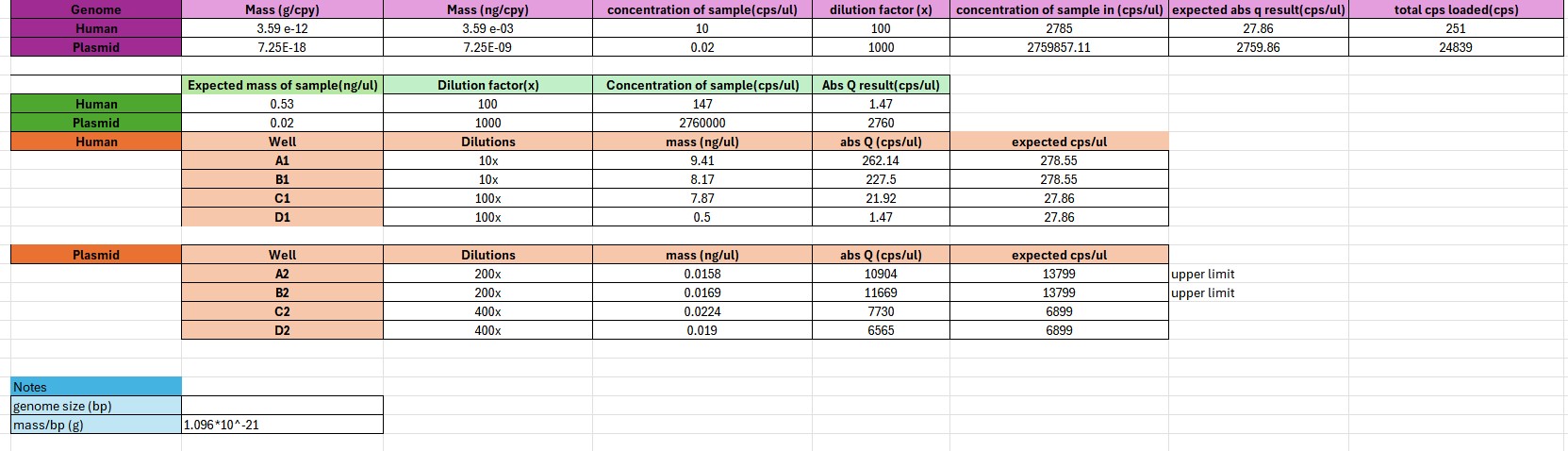
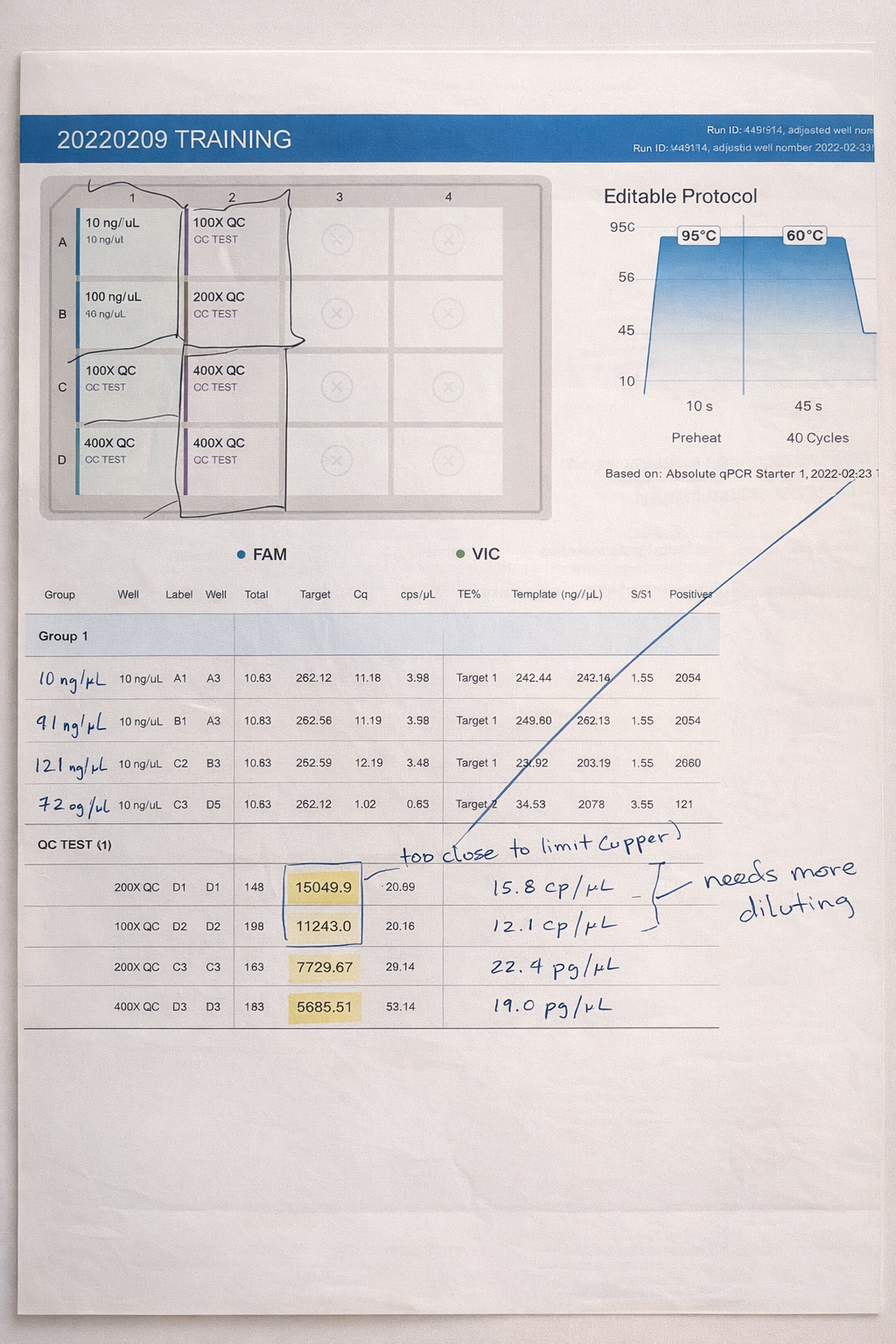
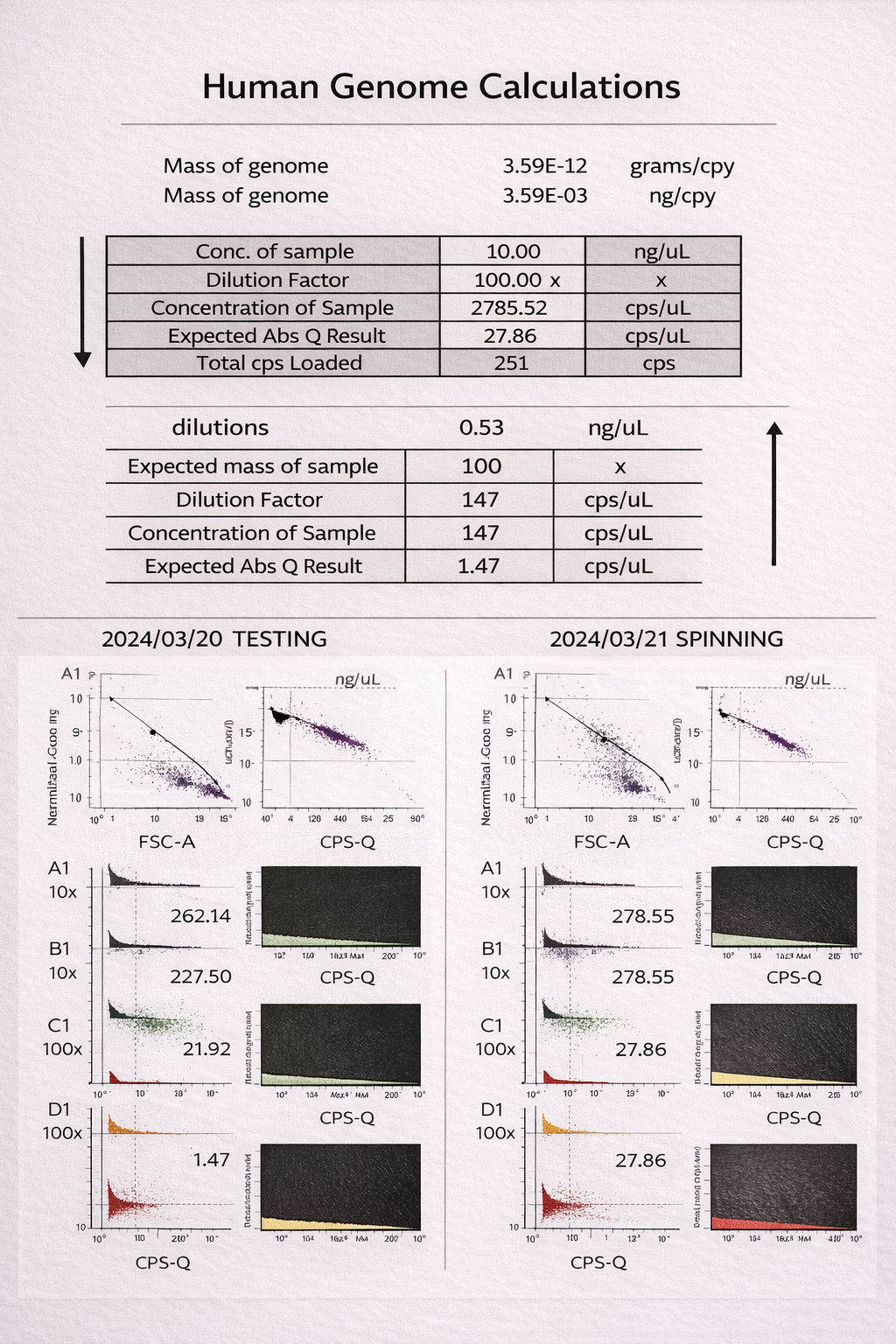
This project focused on the use of digital PCR (dPCR) to achieve highly accurate, absolute quantification of plasmid DNA in drug product formulations used for gene therapy and DNA-based therapeutics. Unlike qPCR, which relies on standard curves and amplification efficiency, dPCR partitions samples into thousands of micro-reactions and directly counts target molecules using Poisson statistics.

By eliminating standard curve–dependent variability and reducing sensitivity to plasmid topology and PCR inhibitors, dPCR provides improved precision and reproducibility—particularly at low copy numbers and in complex formulation matrices. These advantages make dPCR especially well-suited for dose determination, lot-to-lot comparability, and regulatory release testing of plasmid DNA drug products.
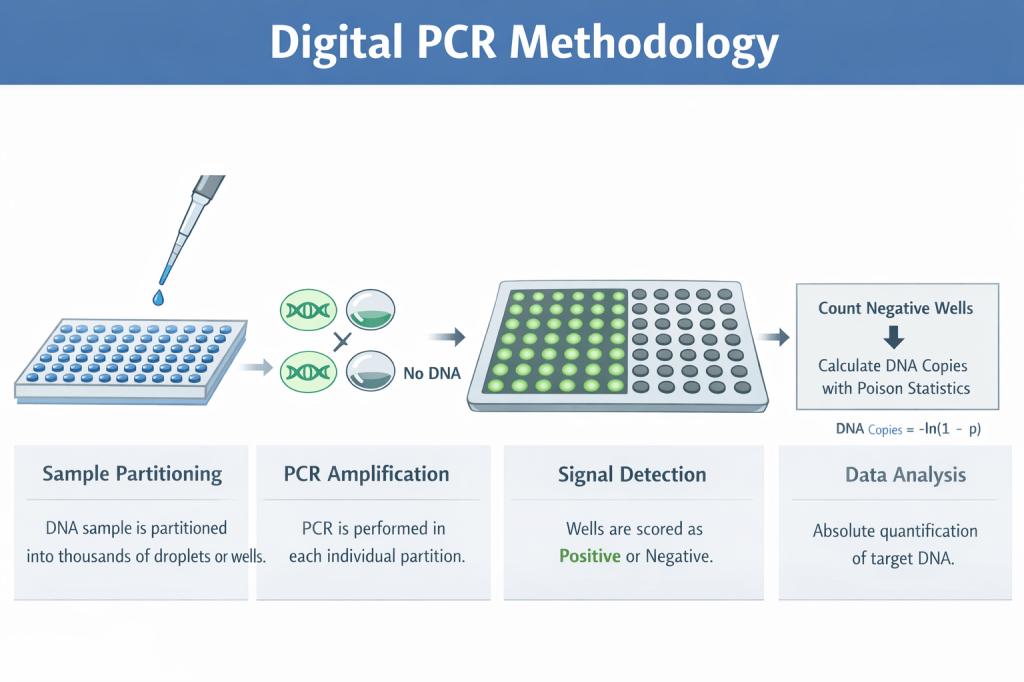
Accurate plasmid quantification is critical for ensuring patient safety, consistent manufacturing performance, and reliable downstream viral vector production. This work highlights dPCR as a robust analytical tool that strengthens both product quality and regulatory confidence in nucleic acid–based therapeutics.